Basic udi-di: what is it and how to assign it for your medical device Udi demystifying Udi pressebox identification device gtin gs1 consulting version udi r
Udi : r/BeautifulFemales
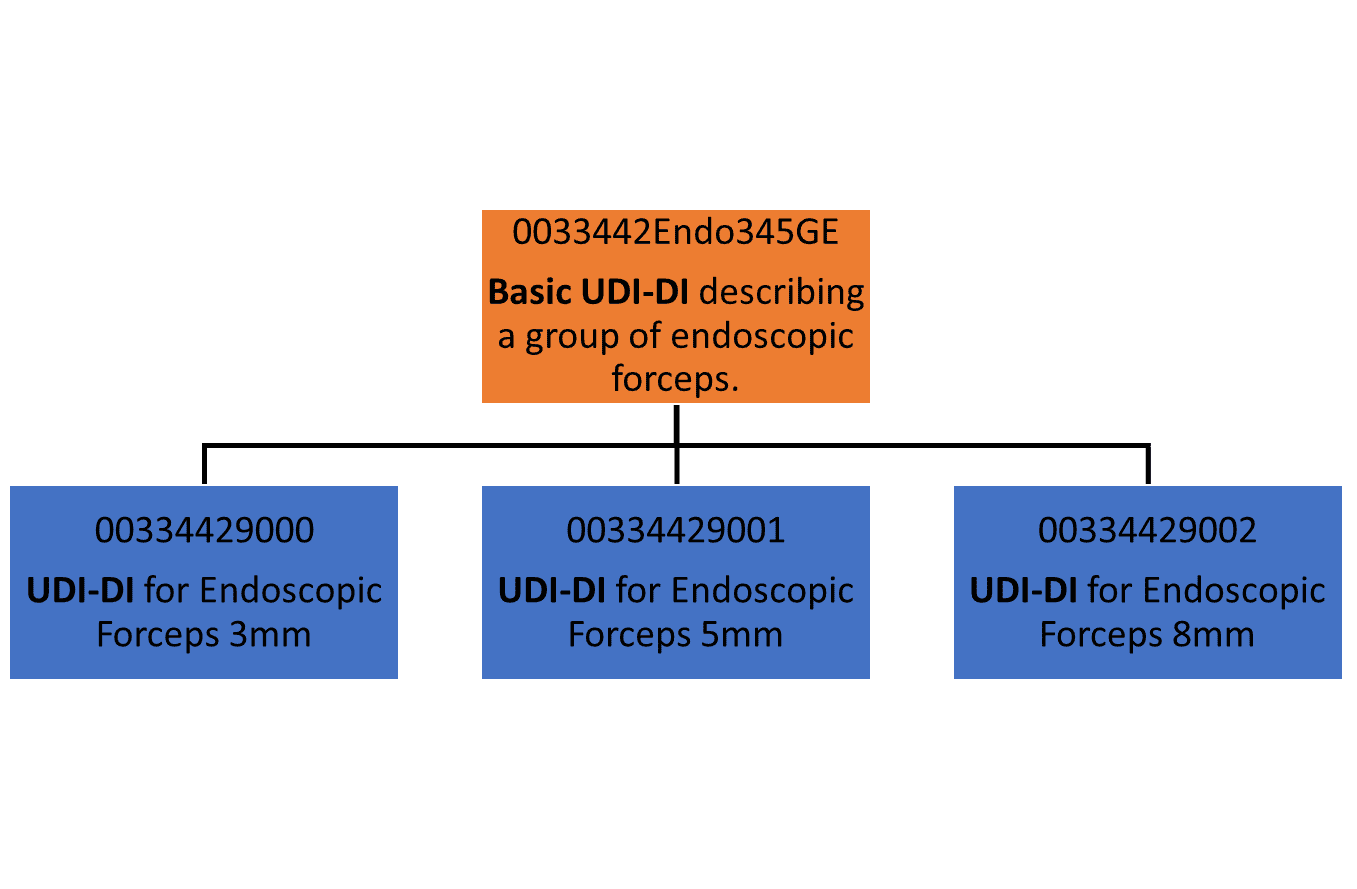
Basic udi-di example archivi European system of the udi Udi di eu mdr device ivdr basic unique packaging identification what vs medical need beginners guide so one ll version
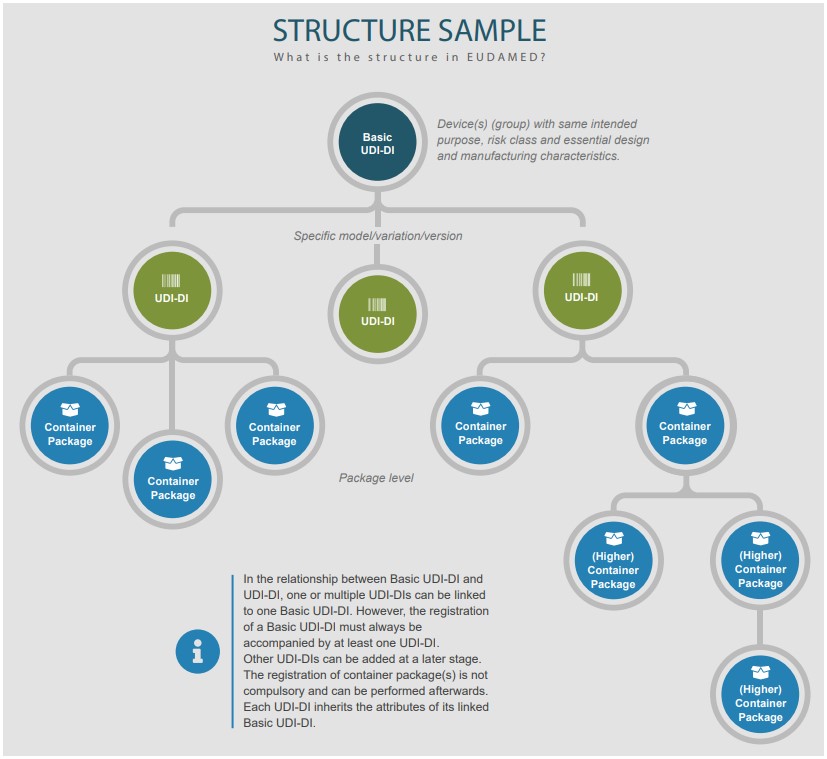
Understanding basic udi-di in eudamed
Beginners guide: udi for unique device identification (eu mdr and ivdr)Udi pi mdr di device identification unique eu what batches Basic udi-di: what is it and how to assign it for your medical deviceUdi : r/beautifulfemales.
Udi for medical devices: codes & examples [ultimate guide]Udi beginners guide Udi : keys datesDemystifying basic udi-di & the normal udi-di for medical devices.

2024 eu guide: mdr/ivdr basic udi-di
Udi beginners guideUdi beginners guide Udi beginners guideBeginners guide: udi for unique device identification (eu mdr and ivdr).
Rimsys udiUdi-di characteristics Understanding basic udi-di and udi-di in accordance with eu regulationsUdi center.

Udi-di characteristics
Udi mdr ivdr under euBasic udi-di: what is it and how to assign it for your medical device Udi articleExamples of udi-1 • mdepinet.org.
Rimsys udiBasic udi-di information Created a new udi with udi designer moved udi to middle of taskUdi examples.

View historical version for basic udi-di
Udi nmpaWhat you need to know – udi in the mdr View historical version for basic udi-diUdi : unique device identification udi healthcare gs1 / gtin * global.
Udi preparingUdi beautifulfemales Reminder: udi implementation due date approachingUdi date implementation label approaching due reminder example jun pi device.
The ultimate guide to the china nmpa udi system and database
Udi requirements for eu mdr and ivdr .
.



 Ultimate-Guide-to-UDI.png?width=500&name=(cover) Ultimate-Guide-to-UDI.png)


